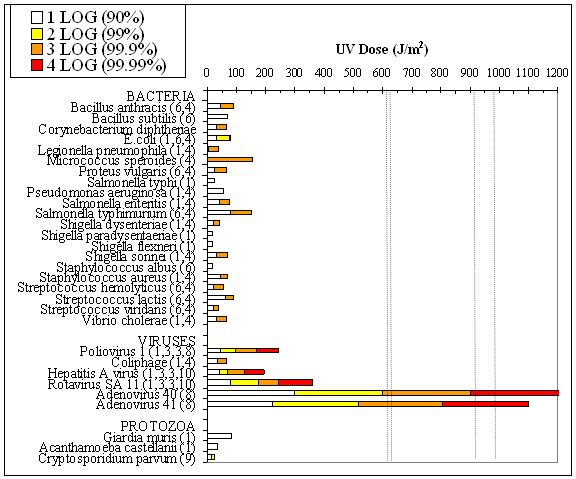
To determine the germicidal effectiveness of each UV-Tube prototype, MS2 bacteriophage was used to determine the average fluence (dose) of UV light at a specified flow rate. First, a fluence-inactivation curve was generated for the MS2 by exposing samples to varied amounts of UV light using a bench-scale quasi-collimated beam apparatus. Water with a UV254 transmittance of 73% was inoculated with MS2 and then passed through the UV-Tube prototype at three flow rates. The log inactivation of the MS2 was compared with the fluence-inactivation curve to determine the fluence (dose). Results are shown below on a plot of inactivation data for pathogens of concern (compiled from various sources). All designs, at the flow rates tested, provide a fluence which exceeds the 400 J/m2, as required by the National Sanitation Foundation standards (NSF/ANSI 55 “Ultraviolet Microbiological Water Treatment Systems”).

Vertical lines correspond with the following test results:
620 J/m2: Large Stainless Steel Lined PVC at 8 L/min
630 J/m2: Medium Pottery at 5 L/min
920 J/m2: Large Stainless Steel Lined PVC at 5 L/min
980 J/m2: Large Concrete at 5 L/min
Three independent bioassay experiments were performed over a period of three weeks using a stock solution of MS2, (Escherichia coli bacteriophage ATCC® 15597-B1) prepared eleven months prior, following propagation steps as described in ISO 10705-1 using an E.coli host (Escherichia coli ATCC® 15597) and stored at 4°C. Berkeley City tap water was treated with a course filter and dechlorinated by passing through an activated carbon filter. The absorbance of the treated water was 0.008-0.013 cm-1 (transmittance 97-98%). MS2 was added to the feed water, such that the concentration entering the disinfection device was 106 PFU/mL. Constant head was maintained in the feed tank and a flow control valve and flow meter were used to produce a constant flow rate to the water disinfection device. Steady-state conditions were established in the device by continuous operation for at least 10 hydraulic residence times. Three 50-mL samples were collected upstream of the device as inlet samples and five 10-mL samples were collected at the outlet. At least one inlet sample preceded and one followed collection of the outlet sample series. MS2 was enumerated in the water samples using the double layer agar method, as first described by Adams. Once sample was added to host culture, less than one minute passed before the mixture was plated and covered to prevent potential photoreactivation.
A quasi-collimated beam apparatus was used to determine the fluence-response curve for MS2 in the feed water. Irradiance at the center of the surface of the sample was measured using a Spectroline® Digital radiometer (DM-254XA, Spectronics Corporation, Westbury, NY) and fluence was calculated taking into account various factors, including sample volume, distance from UV source, path length through water and absorbance, as described by Bolton and Linden. Prior to the first bioassay, a sample of feed water was divided into aliquots that were exposed to a range of fluences: 200, 400, 600, 800 and 1000 J/m2, each with three replicates, to generate a standard curve. For each study, a portion of the feed water was exposed to three fluences, each with 2 replicates, to confirm that response of the MS2 had not changed.
Quasi-collimated beam results, shown below, provided a relationship between fluence and log inactivation of MS2. Quasi-collimated beam results conducted on each of the three testing days were consistent with the standard fluence-response curve. The reciprocal of the correlation given by the data was used to back-calculate fluence based on log reduction of the effluent samples from the disinfection device.
The logarithm of the MS2 inactivation with 95% confidence intervals for the three testing days, each at 5 L/min, are shown below.
| Date | UV254 Absorbance
(cm-1) |
Transmittance at 254nm | No. of Samples | Log Inactivation | Standard Deviation | Mean Equivalent Fluence (J/m2) (95% confidence interval) |
|---|---|---|---|---|---|---|
| 02/27/03 | 0.011 | 98% | 5 | 3.8 | 0.53 | 798 (678-917) |
| 03/07/03 | 0.013 | 97% | 5 | 5.1 | 0.45 | 1133 (1032-1234) |
| 03/12/03 | 0.010 | 98% | 5 | 5.2 | 0.42 | 1155 (1060-1249) |
| Mean | 0.011 | 97% | 15 | 4.7 | 0.79 | 1017 (915-1118) |
The mean MS2 concentration in the inlet was used to calculate the log inactivation for each outlet sample. Inactivation results for the UV disinfection system are summarized in Table 1. It is not clear why a consistently lower inactivation was observed during the first experiment (samples 1-5) compared to the second and third experiments. It is possible that the bulb was dirty due to a longer period of non-use prior to experiment one. Another possibility is that the bulb output was lower; the bulb used for the experiments had been turned on for less than 100 h, and the output is known to fluctuate during this “burn in” period. Experiments are currently underway to quantify the fluctuation in output during the lifetime of the bulbs. Nonetheless, the mean equivalent fluence of 1017 J/m2 is significantly higher than the 400 J/m2 minimum fluence required by the National Sanitation Foundation protocol. For comparison, the fluence provided by the disinfection system was greater than that needed to provide a 4-log inactivation of poliovirus, hepatitis A virus, and rotavirus, as well as a variety of pathogenic bacteria and protozoa. The device would provide a 3-log inactivation of Adenovirus types 40 and 41, which are some of the most UV resistant waterborne pathogens known.